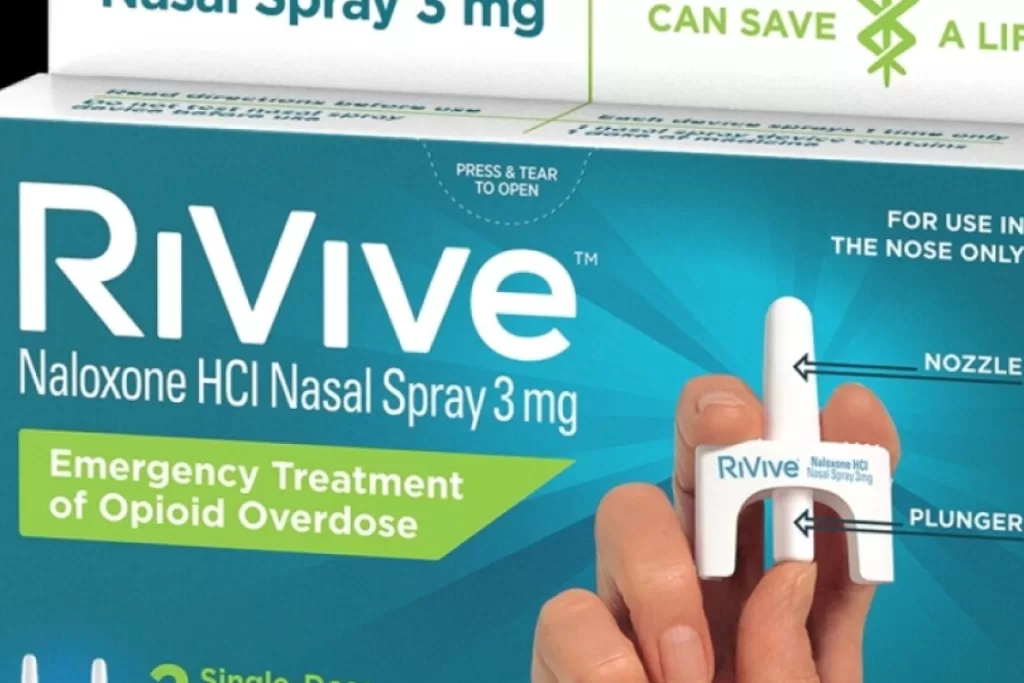
- FDA grants approval for RiVive, a nasal spray version of naloxone, as a prescription-free opioid overdose reversal option.
- Harm Reduction Therapeutics to provide at least 200,000 free doses of RiVive to harm-reduction organizations and states.
- RiVive’s availability offers a timely response to the rising opioid-related overdose death toll in the United States.
The U.S. Food and Drug Administration (FDA) has granted approval for the prescription-free sale of RiVive, the second opioid overdose reversal drug, as announced by its manufacturer, Harm Reduction Therapeutics. This development offers patients another over-the-counter option in the United States, where drug-related overdose deaths have surpassed 100,000 in 2021.
The newly approved drug, RiVive, will be made available early next year, primarily to harm-reduction organizations and state governments. Harm Reduction Therapeutics, a not-for-profit drugmaker, aims to provide at least 200,000 doses of RiVive for free.
RiVive is a nasal spray version of naloxone, a medication that rapidly reverses the effects of opioids, restoring normal respiration, particularly when administered within minutes of the initial signs of an overdose.
Michael Hufford, the co-founder and CEO of Harm Reduction Therapeutics, expressed the company’s commitment to lowering the cost of RiVive or increasing the quantity available for free if additional funding support is received from partners.
Earlier in March, Emergent BioSolutions obtained FDA approval for the first over-the-counter version of Narcan, also a naloxone-based nasal spray.
Harm Reduction Therapeutics has collaborated with contract drug manufacturer Catalent Inc to handle the production of RiVive.
This approval marks a significant step towards combating opioid overdose fatalities, and the availability of RiVive without a prescription offers greater accessibility to a life-saving medication for those at risk.